BURLINGTON, Mass. & New York–( BUSINESS WIRE )–Butterfly Network, Inc. (“Butterfly,” “The Company”) (NYSE: BFLY), a pioneer and leader in semiconductor-based ultrasound devices, programmable cloud software and AI, today announced that it has received approval from the US Food and Drug Administration (FDA) for its fully automated Gestational Age (GA) handheld solution. It marks an FDA-cleared blind sweep ultrasound AI tool for estimating gestational age.
Butterfly Network secures first FDA clearance for blind-sweep ultrasound AI device — a big step for women’s health.
to share
Despite its importance in guiding prenatal care, accurate assessment of gestational age remains a gap in maternal care worldwide. In the United States, nearly half of rural counties lack hospital obstetric services, and communities with socioeconomic barriers have poorer outcomes. Globally, 92% of maternal and newborn deaths occur in low- and middle-income countries – many of which are preventable with early detection and early intervention. In these settings, limited access to ultrasound and trained professionals make it difficult to establish gestational age, especially in emergencies where patients may be unaware or unable to communicate. Early detection enables clinicians to time key prenatal tests, detect complications early, guide intervention, and ultimately improve outcomes for mothers and babies.
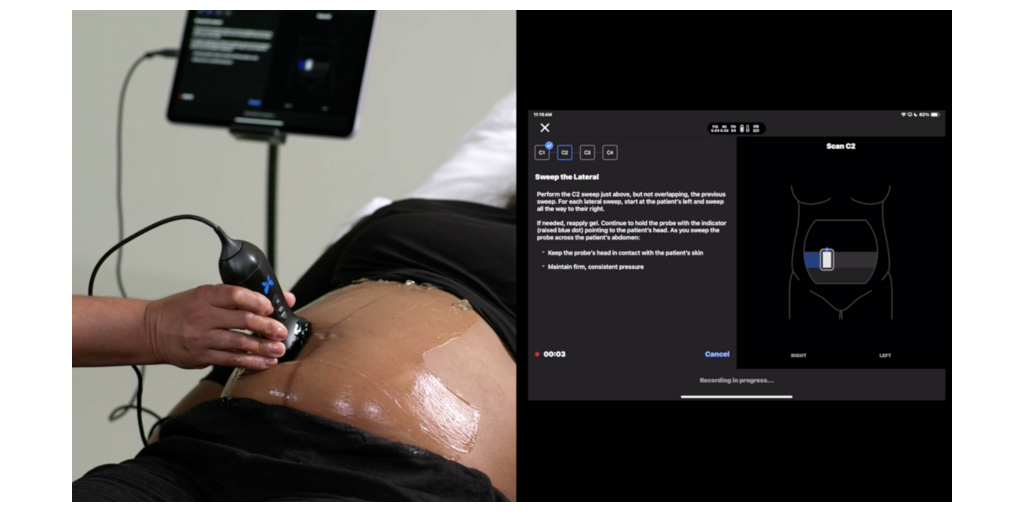
Butterfly’s GA Tool builds on deep learning models developed by Jeffrey Stringer, MD, and team at the University of North Carolina at Chapel Hill, and uses an AI-powered blind sweep method to provide a reliable estimate of gestational age in less than two minutes. It provides quick insights by eliminating the need for biometric measurements.
Trained on more than 21 million images of different patient demographics and care settings, the model provides consistent results in patients between 16-37 weeks performed by a sonographer, equivalent to biometric-based gestational age assessments. The solution is integrated directly into the Butterfly app and can be made available to caregivers worldwide.1
“Improving maternal health outcomes and expanding access to prenatal imaging has become an urgent priority, and Butterfly is proud to be the first in the world to bring this technology to mothers,” said Sachita Shah, MD, Vice President, Global Health, Butterfly Network. “With FDA clearance, we now have an AI-powered tool that can help transform maternal health, both around the world and here in the United States, by addressing the chronic gap in access to timely, reliable imaging and enabling earlier, more informed care.”
Steve Cashman, Butterfly’s chief business officer, commented, “The GA tool is designed to address three critical use cases: enabling rapid decisions in emergency settings when gestational age is unknown; expanding access in countries where maternal and neonatal mortality are high; and improving prenatal ultrasound access where ultrasound access can be improved for more patients in rural areas of the United States. It’s about helping clinicians act faster, earlier and more confidently, in critical moments and in any setting.
Butterfly’s GA tool has already been deployed in Malawi and Uganda, and is made possible with a grant from the Gates Foundation. The FDA clearance enables a fast track to expand to additional regions in sub-Saharan Africa and comes at an opportune time in the United States, as federal rural health transformation initiatives encourage investment in expanding access to care. Organizations interested in partnering with Butterfly on maternal health programs can learn more here: https://www.butterflynetwork.com/global-health
About the butterfly network
Butterfly Network, Inc. (NYSE: BFLY) is a healthcare company driving the digital revolution in medical imaging with its proprietary ultrasound-on-a-chip™ semiconductor technology and ultrasound software solutions. In 2018, Butterfly launched the world’s first handheld, single-probe, whole-body ultrasound system, the Butterfly iQ. The iQ+ followed in 2020, and the iQ3 in 2024, each using Moore’s Law to improve processing power and performance. The iQ3 received Best Medical Technology at the 2024 Prix Galien USA Awards, a prestigious honor and one of the highest awards in healthcare. Butterfly’s innovations have also been recognized by the Ferris 50, TIME’s Best Inventions and the Fast Company’s World-Changing Ideas, among other achievements.
Butterfly combines advanced hardware, intelligent software, AI, services, and education to optimize affordable, accessible imaging. Clinical publications show that this handheld ultrasound probe, paired with Compass™ enterprise workflow software, can help hospital systems improve care workflow, reduce costs and improve provider economics. With a cloud-based solution that enables care anywhere through next-generation mobility, Butterfly aims to democratize healthcare by addressing global healthcare challenges. Butterfly devices are commercially available to trained healthcare professionals in areas including, but not limited to, Africa, Asia, Australia, Europe, the Middle East, North America, and South America; For more information on available countries, visit: https://www.butterflynetwork.com/choose-your-country.
1 Butterfly presets, drawing modes and features are not all available everywhere. Check availability, subscription type, and software version in your country.
#Butterfly #Network #Secures #FDA #Clearance #Blind #Sweep #Ultrasound #Device #Marks #Major #Advance #Womens #Health