I’ve written recently about nitazines ( here and here ), a class of deadly synthetic opioids that have started showing up in the street drug supply over the past few years. Just what we don’t need. These bad boys are extraordinarily potent—often more so than fentanyl—and easy to make using common laboratory chemicals. In other words, another street drug.
And the band played
Recently, Jeff Singer and I wrote about another chemical disaster: Cyclophene. Different structure, same form – very dangerous. Yet another opioid that can be synthesized from a new precursor set, prevents controls that eventually begin to interact with fentanyl.
None of this was surprising. This is the same cycle we have seen for decades. Crash with one drug, and another—stronger, cheaper, and often more dangerous—takes its place. The prescription opioid gave way to heroin. Heroin gave way to fentanyl. Fentanyl gave way to nitazine. Now something else is already waiting in the wings.
Call it the iron law of Prohibition: the harder the enforcement, the harder the drug.
But every so often, there’s a pleasant surprise around the corner.
In this case, it’s an opioid that—at least in early studies—treats pain better than other, more powerful opioids. And I’ll be damned if this isn’t a Nietzschean analog.
It may even represent a rare case of escaping the iron law of prohibition, thanks to a 36-page opinion. nature– an almost superhuman effort by 44 (!) scientists across multiple research centers, over 30,000 words (!again) – a task I’d happily delegate to someone else.
Here we identify a novel MOR agonist with supramaximal intrinsic efficacy and a unique pharmacological profile that produces effective analgesia in mice with minimal adverse effects.
Gomez, JL, Ventriglia, EN, Frangos, ZJ et al. A µ-opioid receptor agonist analgesic with minimal adverse effects. nature (2026). https://doi.org/10.1038/s41586-026-10299-9
Same goal, more or less
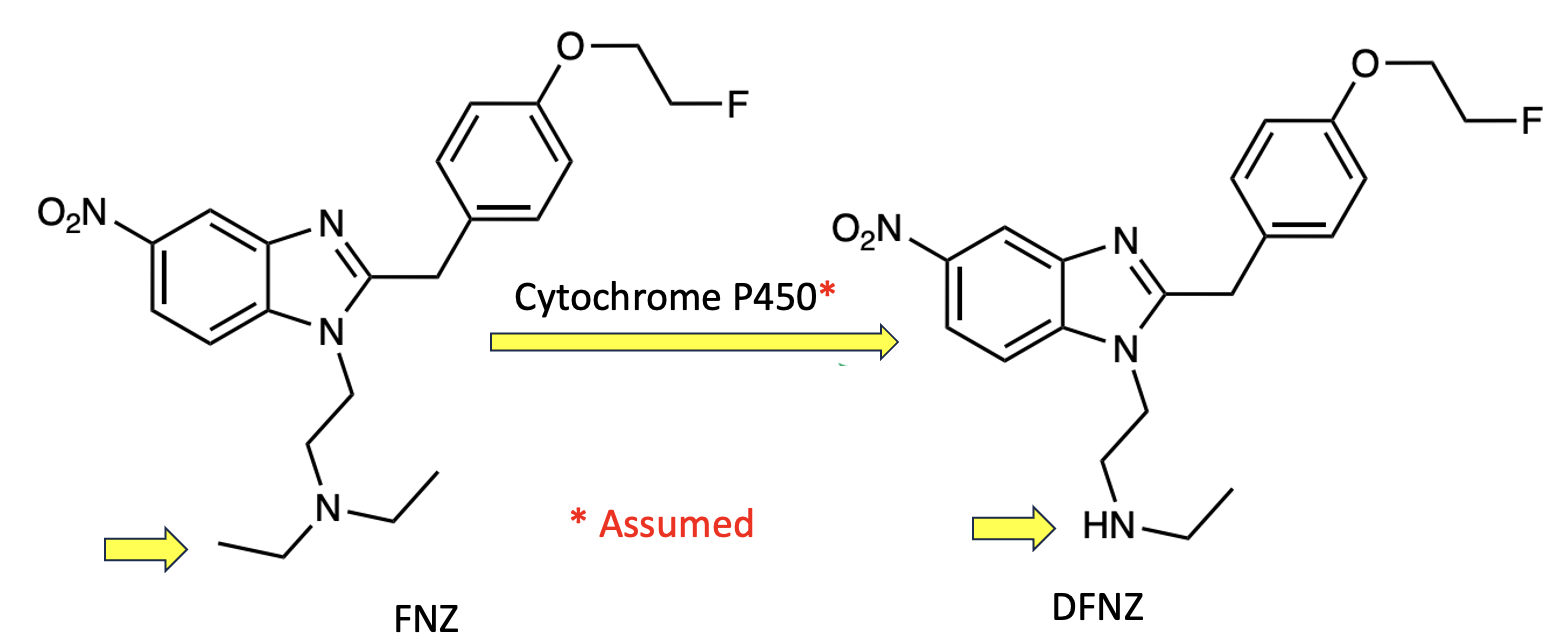
The most important compound is called DFNZ (N-desethyl-fluornitazene), a derivative of the nitazene class (Figure 1). Intuitively, there is nothing certain about it – quite the opposite. Nitasines are among the most powerful opioids known and began appearing in the US illicit drug supply around 2020, with entirely predictable results.
Figure 1. N-Desethyl-fluornitrazene (DFNZ), an active metabolite formed by oxidative deacetylation of fluornitrazene (FNZ), presumably by one of the CYP enzymes in the liver. Yellow arrows indicate the site of metabolism.
DFNZ targets the same target as morphine and fentanyl – the µ-opioid receptor. In laboratory tests, it behaves as a “superagonist,” meaning it can activate the receptor even more strongly than standard opioids (Table 1).
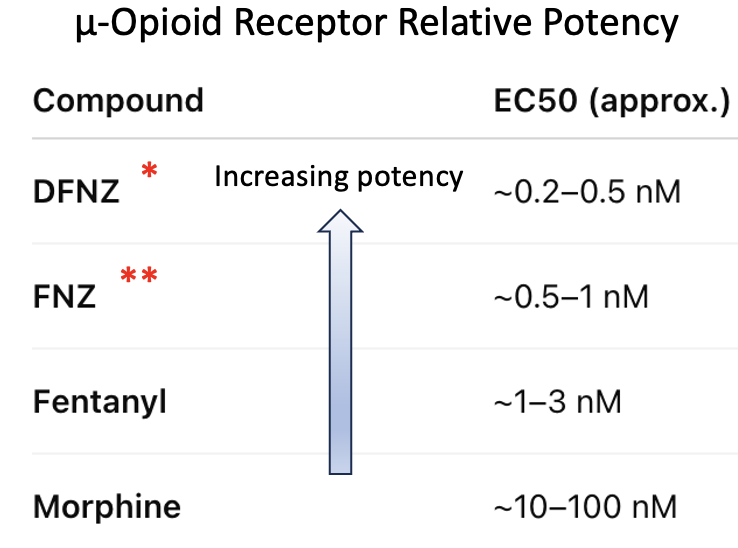
Table 1. Relative potencies of selected opioids. Both natazins are strong mu-agonists. *FNZ, the drug administered, has a short half-life. ** DFNZ, the active metabolite of FNZ, “works”.
So far, nothing new, but…
Closure is just the starting point. What matters is what happens next. This is where DFNZ starts to show its strengths.
It relieves pain – without the usual stuff [1].
At least in a mouse study.
Now things get weird. good weird
The parent compound (FNZ) barely sticks around. PET imaging shows that it is only in the brain for a few minutes. That should be the end of the story.
It is not. The pain lasts for hours. With opioids, the rule is simple: the drug in the brain, the effect. The drug is gone, the effect is gone. not here.
What happens is that FNZ quickly turns into something else—DFNZ. And DFNZ stays on long enough to do real work.
In other words, the drugs given to the mice are not the drugs that matter.
Opioids, dopamine, and other assorted suspects
If opioids are responsible for a signature beyond respiratory depression, it’s addictive—and it’s largely through dopamine. Many addictive drugs stimulate a rapid, rapid burst of dopamine in the brain’s reward circuit. These spikes act as a kind of teaching signal: that’s it, do it again.
DFNZ doesn’t – at least not in the same way. It still increases dopamine, but more slowly and less dramatically. Instead of sharp spikes, the signal is smoother and more consistent. This difference is reflected in behavior. Animals will take the medicine, but when it is removed, they stop. There is little evidence of persistent drug seeking—the kind you almost always see with traditional opioids. Without these rapid dopamine spikes, the brain doesn’t seem to receive the same reinforcement signal. This is not a weak reward signal. This is another one.
The bottom line. maybe
What makes this paper interesting and potentially groundbreaking [3] Not just compound. This is what it suggests.
For a long time, the assumption was that if you strongly activate the µ-opioid receptor, you get the whole package: pain relief, euphoria, respiratory depression, and addiction. You can tuck around the edges, but you can’t really pull them apart.
DFNZ suggests this may be wrong, at least in the “rodent world”.
Perhaps the pain cannot be resolved by quitting opioids, but by using a different medication.
This is a great idea. It is also a long way from fixed. Everything here is in rats, but it’s still a very powerful opioid with properties that have defied previous opioid research. This is hardly a remarkable thing.
If these findings hold up—still a big if—they point to something we haven’t seen before: an opioid that relieves pain without fully activating the biological machinery that makes opioids so dangerous.
It’s really something. But, for now, keep your legs crossed. This discovery may be too good to be true. Time (and mice) will tell.
Finally, let’s close with some wisdom from my frequent writing colleague, Dr. Singer. What would have happened if the DEA had made natazin a schedule I drug?
Fortunately, regulators didn’t panic and jump at the timing of Netizen’s release in 2019. We have seen what happens otherwise: Schedule I has buried psychedelic research for fifty years, and we are only now uncovering its therapeutic potential.
He has a point.
Note:
[1] “Assets” include respiratory depression, tolerance, dependence, withdrawal, and reinforcement of drug-seeking behavior.
[2] The fluorine atom isn’t just decorative—it allows the compound to be labeled with fluorine-18 for PET imaging, so researchers can track where it goes in the brain in real time.
[3] I left out a lot. Given the length and complexity of this paper, I really had no choice.
#supropiod #treat #pain #opioids #Nature #paper